Indicators
indicators determine whether a solution is an acid or a base
by turning a different color if there is a change in the pH
by turning a different color if there is a change in the pH
Phenolphthalein
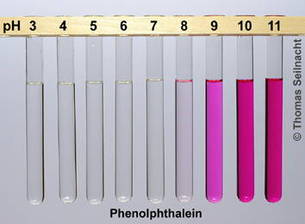
in a basic solution phenolphthalein turns purple
in a neutral solution phenolphthalein is colorless
and in an acidic solution phenolphthalein is also colorless
WHY IS IT USEFUL: the best indicator to use in titrations because it is the most accurate when trying to find the same concentration of an acid and a base solution
in a neutral solution phenolphthalein is colorless
and in an acidic solution phenolphthalein is also colorless
WHY IS IT USEFUL: the best indicator to use in titrations because it is the most accurate when trying to find the same concentration of an acid and a base solution
Litmus Paper
(blue and red)
(blue and red)
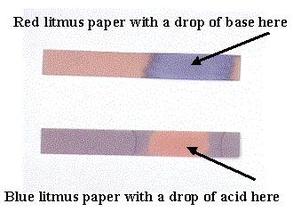
bases turn red litmus paper blue (acidic and neutral solutions do not change the color of the litmus paper)
acids turn blue litmus paper red (basic and neutral solutions do not change the color of the litmus paper)
WHY IS IT USEFUL: helps to determine whether or not an unknown solution is either an acid or a base
acids turn blue litmus paper red (basic and neutral solutions do not change the color of the litmus paper)
WHY IS IT USEFUL: helps to determine whether or not an unknown solution is either an acid or a base
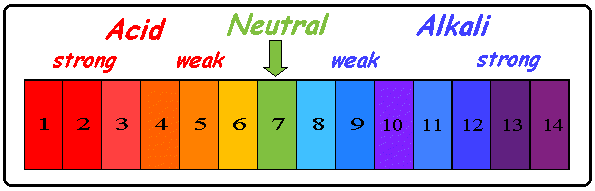
Universal Indicator
(comes in liquid and paper forms)
(comes in liquid and paper forms)
|
able to turn a certain color depending on the solution, and you are able to associate the color it turns with a certain pH number
WHY IS IT USEFUL: helps to determine whether an unknown solution is acidic or basic as well as it's pH number |
If you've mastered all the concepts ranging from the theories of acids & bases to indicators, move on to...