Strength vs. Concentration of Acids and Bases
|
|
concentration is the amount of the acid or base per unit volume (liter)
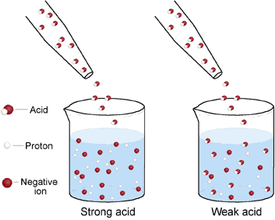
strength of an acid is dependent on the concentration of hydrogen ions, and the ability of the acid to lose hydrogen ions, meaning it’s also able to lose it’s concentration of hydrogen ions
strength of a base is dependent on the concentration of hydroxide ions, and the ability of the base to lose hydroxide ions, meaning it’s also able to lose it’s concentration of hydroxide ions
|
Molarity
|
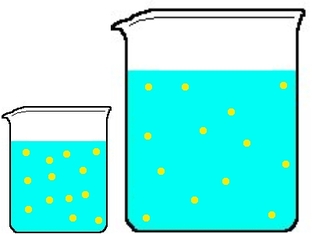
although these beakers hold different amounts of the same solution, their concentration is the same thus their molarity is the same
|
Molarity (M): the molar concentration of a solution, expressed as the number of moles of solute per liter of solution
Some people confuse Molar concentration with strength, believing that the more concentrated a solution is, the stronger the solution. However, this belief is WRONG! In actuality the RIGHT statement is that the more concentrated a solution is, the higher the amount of acid or base per unit volume. |
moles are the amount of chemical substance
M = number of moles/number of liters
1000 cm3 = 1 liter
1 cm3 = milliliter
M = number of moles/number of liters
1000 cm3 = 1 liter
1 cm3 = milliliter
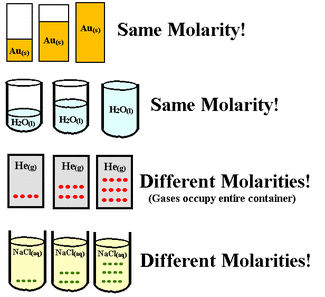
Explaining the Imagethe beakers full of Au have the same molarity with one another because they all have the same concentration, this is also true for the beakers of H2O
BUT for the beakers full of He they have a different molarity because they all have different concentrations, this is also true for the beakers NaCl |
Understand strength, concentration, and molarity?