Titrations
Why do you preform a titration?
To find the unknown concentration of a sample by using a reagent with a known concentration.
To find the unknown concentration of a sample by using a reagent with a known concentration.
Types of Titration
|
rough titration: only trying to gain a sense of how much acid is needed in order to neutralize the base, or how much base is needed to neutralize the acid
|
exact titration: trying to measure the exact amount of how much acid is needed in order to neutralize the base, or how much base is needed to neutralize the acid
|
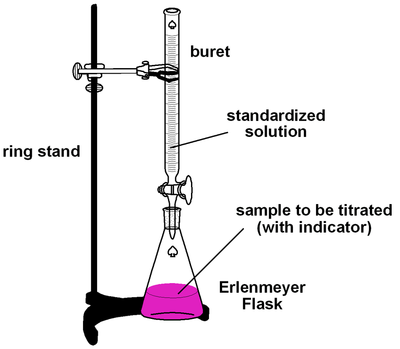
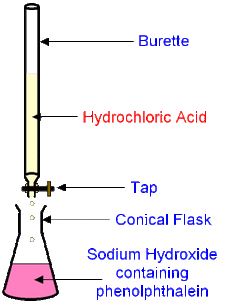
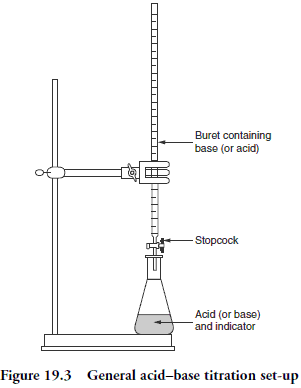
above the apparatus used in titrations is shown
(below there is a gallery with more pictures of the titration process)
(below there is a gallery with more pictures of the titration process)
How to Perform a Titration
in this titration we will be using HCl (acid) and NaOH (base) and if everything goes well,
the neutralization reaction of the HCl + NaOH --> NaCl (salt) + H2O (water) will occur
the titration process might seem complicated,
but it is quite simple (this is just a through procedure,
the process is not as daunting as it seems)
the neutralization reaction of the HCl + NaOH --> NaCl (salt) + H2O (water) will occur
the titration process might seem complicated,
but it is quite simple (this is just a through procedure,
the process is not as daunting as it seems)
|
Procedure
1. put on safety goggles and put away any dangling items
2. gather all your materials 3. label your beakers “A” (acid) and “B” (base) 4. wash beakers “A” and “B”, dry and get rid of all the water 5. pour AT LEAST 75cc of HCl into the beaker labeled “A” and pour AT LEAST 75cc of NaOH into the beaker labeled “B” 6. add water to the burette, and clean out the burette (twice), then clean the burette with HCl in order to prevent the HCl from being diluted 7. attach the burette to the clamp stand and put the funnel at the top of the burette |
Materials
- clamp stand
- two smallish beakers (size doesn’t matter because they are just containers, doesn’t matter if they are plastic or glass) - one conical flask, Erlenmeyer flask - one volumetric pipet - one buret - one funnel - phenolphthalein (indicator) - Hydrochloric Acid (acid) - Sodium Hydroxide (base) - safety goggles (one per person) - sink or some kind of water supply where you can wash certain materials |
8. pour HCl into the burette, enough so that you will be able to measure AT LEAST 10cc of HCl
9. record the initial amount of HCl in the burette before the titration
10. take the funnel out and place it somewhere that is clean so that no contamination occurs
11. wash the flask out with water, you DO NOT have to dry the flask (answer to this question is on this page)
12. add water to the volumetric pipet, and clean out the volumetric pipet, then clean the volumetric pipet with NaOH in order to prevent the NaOH from being diluted
13. measure EXACTLY 10cc of NaOH using the volumetric pipet (the meniscus should be on the mark for 10cc)
14. add the 10cc of NaOH to the cleaned flask, be sure to NOT get chemicals on the sides of the flask (answer to this question is on this page)
15. add 3 drops of phenolphthalein to the NaOH
16. place the flask with the NaOH and phenolphthalein inside of it at the base of the clamp stand
9. record the initial amount of HCl in the burette before the titration
10. take the funnel out and place it somewhere that is clean so that no contamination occurs
11. wash the flask out with water, you DO NOT have to dry the flask (answer to this question is on this page)
12. add water to the volumetric pipet, and clean out the volumetric pipet, then clean the volumetric pipet with NaOH in order to prevent the NaOH from being diluted
13. measure EXACTLY 10cc of NaOH using the volumetric pipet (the meniscus should be on the mark for 10cc)
14. add the 10cc of NaOH to the cleaned flask, be sure to NOT get chemicals on the sides of the flask (answer to this question is on this page)
15. add 3 drops of phenolphthalein to the NaOH
16. place the flask with the NaOH and phenolphthalein inside of it at the base of the clamp stand
|
for a rough titration you would want to follow these steps next:
17. open the burette and let the HCl flow into the conical flask all the while swishing around the solution in the burette
18. once the phenolphthalein turns colorless close the burette (try and close the burette as soon as the solution turns colorless) 19. record the final amount of HCl in the burette after the titration, subtract the final amount of HCl from the initial amount (the closer to 10cc, the more accurate your results) |
however for an exact titration you would want to follow these steps next:
17. open the burette and let the HCl flow into the conical flask all the while swishing around the solution in the burette
18. once 7cc of HCl has been added to the flask, close the flow on the burette enough so that the HCl is dripping (but make sure not to fully close the burette) 19. once the phenolphthalein turns colorless close the burette (try and close the burette as soon as the solution turns colorless for more accurate results) 20. record the amount of HCl in the burette after the titration, subtract the final amount of HCl from the initial amount (the closer to 10cc, the more accurate your results) 21. clean out all your materials 22. repeat as many trials as possible using the same directions found in steps 1-16, and then steps 17-21 in the exact titration section |
We're not done with titrations just yet...